 
Figure 5 (silver): Courtesy of /./photogallery/ģ. For example, gold and silver have different atomic masses and different properties.įigure 4 (Gold): Courtesy of Chris Ralph that released this image into the public domain. All atoms of an element are alike in mass and other properties, but the atoms of one element differ from all other elements. Figure 3: Courtesy of Yzmo on Wikimedia commons, available under Creative Commons-Share Alike 3.0 UnportedĢ. :max_bytes(150000):strip_icc()/GettyImages-845813912-fd50c340e6dd4b7f9fd639ca80a0ebd7.jpg)
The following sections discuss this further. The black area around the nucleus represent the electron cloud. The purple and red dots represent the neutrons and protons in the nucleus. Atoms can neither be created nor destroyed. Each chemical element is composed of extremely small particles that are indivisible and cannot be seen by the naked eye, called atoms. Figure one as a whole constructed by Jessica Thornton using Microsoft Word and Preview (UCD). Scientists did not take into account the gases that play a critical role in this reaction. However, the illustration below shows that the burning of word does follow the law of conservation of mass. From this observation scientists concluded that mass had been lost. 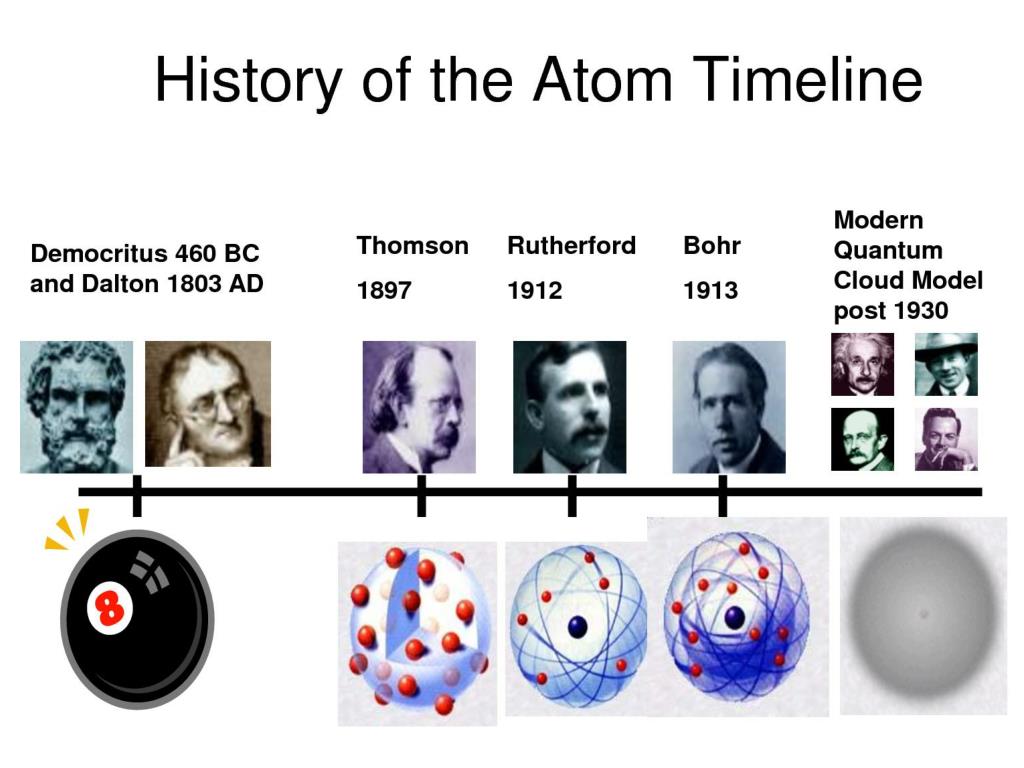
If this law was true, then how could a large piece of wood be reduced to a small pile of ashes? The wood clearly has a greater mass than the ashes. Historically, this was a difficult concept for scientists to grasp. The law of conservation of mass was formulated by Antoine Lavoisier (1743-1794) as a result of his combustion experiment, in which he observed that the mass of his original substance-a glass vessel, tin, and air-was equal to the mass of the produced substance-the glass vessel, “tin calx”, and the remaining air. The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction in other words, mass is conserved. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
